- About Us
About RSIS International
Publication Method
Conference
Contact Us
- Journals
- Conference Proceedings
Days
Hours
Minutes
Seconds
Submission Deadline
Human Immunodeficiency Viral Treatment in a Tertiary Facility Concerns on Prevalence and Predictors of Viral Load Non-Suppression among Children (0-9) and Adolescents (10-19) Attending Clinic, South-West, Nigeria: A Retrospective study
Principal Investigator:
Dr. Ahmed Kayode AbdulMumin. Department of Community Medicine and Primary Care, Federal Medical Centre Idi-Aba, Abeokuta, Ogun State.
Other Authors:
Dr. MorufatOgundeyi. Department of Paediatrics. Federal Medical Centre, Idi-Aba, Abeokuta. Ogun State
Dr. Solomon Olorunfemi Olorunsaiye. Department of Community Medicine and Primary Care, Federal Medical Centre Idi-Aba, Abeokuta, Ogun State.
Oluwadamilola Mary Oladunjoye. Department of Community Health, College of Health Sciences. Obafemi Awolowo University (OAU), Ile-Ife, Osun State. Nigeria
Dr. Adediran Saheed Adekunle. Department of Paediatrics. Federal Medical Centre, Idi-Aba, Abeokuta. Ogun State
Corresponding Author:
Dr. Solomon Olorunfemi Olorunsaiye
Department of Community Medicine and Primary Care
Federal Medical Centre, Idi-Aba, Abeokuta. Ogun State
Background: Despite significant progress made towards achieving theWHO-2030 target particularly African regions, viral load non-suppression (VLNS) especially among children and adolescent remains a substantial challenge in resource limited countries. The introduction HAART has led to a remarkable decrease in global HIV infections, however, the burden of VLNS persists poses a troubling threat to the achievement of the WHO goal at record time.
Objective: This study aims to assess the proportion of virological failure and identify predictors of VLNS among children and adolescents receiving antiretroviral treatment (ART) at the Federal Medical Centre, Abeokuta, Southwest Nigeria.
Methods: A retrospective cross-sectional analysis of clinical data from pediatric patients aged 0-19 years who attended the Federal Medical Center Abeokuta between January 2021 and December 2023 was conducted. The study focused on both patients with viral load ≥ 1000 copies/ml and those with viral load suppression (≤1000 copies/ml).
Results:The mean age was 15.76 ± 3.12 years and majority 41 (30.4%). In 2021, 19 (14.1%) had unsuppressed viral load (VL ≥ 1000 copies/ml). In 2022, unsuppressed viral load was 12 (8.9%) and in 2023, unsuppressed viral load was 16 (11.9%). Compliance to ART was significantly associated with VLNS (P-value-0.025) for year 2021, 2022 and 2023 respectively.A multivariate regression analysis of predictors of Viral Load Un-suppression showed that across the 3-years under review – compliance to ART was a strong predictor of non-suppression.
Conclusion: In spite of resources and gained made in HIV interventions in achieving viral load suppression, a substantial population of children and adolescent still remain virally unsuppressed; 2021 [14.1%], 2022 [8.9%] and 2023 [11.9%] respectively. This study showed that efforts to achieve UNAIDS 2030 is achievable if compliance to ART can be sustained [p-value- 0.025].
Keywords: HIV, Viral-Load-Non-Suppression, Children, Adolescents, Antiretroviral Therapy, South-west, Nigeria.
Human immunodeficiency virus (HIV) remains a major public health problem, particularly in sub-Saharan Africa, where the burden of paediatric HIV is disproportionately high.[1]The Joint United Nation Programme on HIV/AIDS (UNAIDS) estimated that 39.9 million people are living with HIV/AIDS 2023[1] with children and adolescents representing a significant yet vulnerable subset of this population. The introduction of Highly Active Antiretroviral Therapy (HAART) has resulted in a 39% reduction in global HIV infection by 2023 compared to 2010,including a more significant 73% reduction among children aged 0-14.Sub-Saharan African achieved the steepest reductionresulting ina markedimprovement in the quality of life for PLWH.[2]However, despite these achievements, viral load non-suppression (VLNS) continues to pose a major challenge, particularly among children and adolescents.
TheWorld Health Organization (WHO) 95-95-95 targets aim to achieve viral suppression in 95.0% of HIV-positive patients on sustained HAART by 2030. According to the UNAIDS Country report for 2021, in Nigeria about 90% of all people living with HIV knew their status with 90% receiving HAART of which 87% achieved viral suppression in 2021.[1]While Nigeria has made significant improvement in its 95-95-95 target, children and adolescents are lagging behind their adult counterparts.[3] Achieving viral suppression is crucial, not only to improve clinical outcome but also to reduce the emergence of drug-resistant strains and to achieve the concept of “U=U” – that is people with undetectable/suppressed viral load having a near-zero risk of transmitting HIV.[1]
Virological failure (VF) is defined as a plasma viral load that exceeds 1000 copies/ml in an HIV patient following two consecutive viral load measurements 3-months apart, despite adherence to HAART for at least 6 months.[4] Viral load (VL) monitoring for all patients on HAART is the most accurate available measure of the effectiveness of treatment response, a key in diagnosing treatment failure.[1] Some associated factors with VF include poor adherence [5], inadequate dosing, and viral resistance.[6] However, antiretroviral therapy (ART) treatment failure has emerged as a major problem in paediatric HIV patients on HAART.[7]Bernheimer JM et al reported that paediatric ART treatment failure rates were 19.3% to over 32% in the poor resource countries including sub-Saharan Africa.[8] With factors such as lack of access to testing, poor adherence [5], inadequate dosing, viral drug resistance and socio-economic challenges contributing to VLNS among children and adolescents.[6][7]These studies highlight the need for rigorous monitoring of viral load trends to assess the effectiveness of HAART.
Despite the advancement of the HIV treatment program in Nigeria, the factors associated with VLNS among children and adolescent are not well understood, notwithstanding its critical importanceof attaining undetectable or suppressed viral status for this vulnerable population. Studies from other resource-limited sub-Saharan African settings suggest that socio-economic factors, such as parental unemployment status, and individual factors like medication adherence, delayed diagnosis and initiation of ART plays an important role in determining treatment outcomes among this population. However, there is paucity of information from the Nigeria HIV program particularly regarding the predictors of VLNS among children and adolescents.[5]
Understanding the factors associated with Viral Load Non-Suppression (VLNS) in treatment-experienced children is critical to accelerating progress toward ending AIDS by 2030.[9] Martin M. Mulinge et al emphasized the need to encourage monitoring of national viral load trends especially among the paediatric population is critical in determining whether the VLNS is increasing, decreasing, or remaining constant in order to assess the efficacy of cART regimens in controlling the paediatric HIV pandemic.[10]A study among adult Ethiopians accessing showed that prevalence of unsuppressed viral load was 20.3%.[11] Viral load unsuppressed among children were predicated on multifaceted factors, some due to caregivers and parental factor.[11]Owusu et al. in Ghana found that subjects whose parents were unemployed had 5.4 chances of virological failure compared to those with employed parents. [12, 13] Another multivariable analysis, showedindependent predictors of HVL non-suppression were older age at ART initiation and poor medication adherence.[13]
The benefits of viral load suppression cut across immunological and clinical components and should be the driving factors in cases where viral load unsuppressed have been identified in clinical practise. Nelson LJ. et al [14] emphasised the significance of viral load monitoring as a means to identifying immunological and clinical improvement among children and adolescent HIV treatment failure. In addition, at the individual patient level, continued viral suppression can stop the appearance of drug-resistance mutations and reduce the occurrence of failure seen clinically. Again, part of the advantage of viral load monitoring is that patients who succeed in achieving virological suppression early using first-line ARV regimens would stay longer in such regimens, have a reduced risk of acquiring infections that occur opportunistically and face a lower mortality rate.[15]
In a study on viral load non-suppression among adolescent, Joel Maena et al reported that sub-Saharan Africa lags far behind with adolescents carrying the burden of viral-load non-suppression.[16] A UNAIDS report corroborated the fact that adolescents and children, especially young women and girls in sub-Saharan Africa, continue to be disproportionately affected, contributing the largest part of the overall HIV non-suppression prevalence of 47%.[17] Additionally, non-suppressed adolescents and children alike are more prone to HIV-related morbidity and mortality that could be circumvented through IAC and support. Also, IAC mitigates subpar adherence that is common among adolescents and leads to drug-resistant HIV strains, which require expensive second-line or third-line treatments.[18]
In 2019 the Ghana AIDS Commission identified a number of factors leading to viral non-suppression among patients on ART including; being an adolescent/children, nutritional scarcity, income and other economic factors.[19] Similar study in Nigeria on factors associated with Viral load non-suppression discovered that males had a 9% greater likelihood of being non-suppressed. Age groups below 60+ had a higher likelihood of a non-suppressed VL, with the highest in the 0–14 age group. Also, clients enrolled at tertiary and secondary level facilities had the greatest likelihood of a non-suppressed VL.[20] Adedoyin A. et al in Southwest, Nigeria reported certain socio-demographic factors associated with viral load non-suppression which include non- adherence to drugs, the psychological effect of the disease, side-effects of medications, forgetfulness, and combination of anti-retroviral drugs with local herbs and alcohol were the assumed major reasons for viral non-suppression among the patients in this study.[21]
This study, therefore, assess the proportion of virological failure and predictors of viral load non-suppression among adolescent and children accessing antiretroviral treatment in Federal Medical centre, Abeokuta, Southwest, Nigeria. The study was borne out of the need to achieve viral suppression among adolescent and children and by so doing reduce morbidity and mortality associated with poor viral load non-suppression and the attendant social and psychological consequences.
Study Area
The study was conducted at the HIV Clinic of the Federal Medical Centre Abeokuta (FMCA). The HIV clinic was established in 2006, managed by the Department of Community Medicine and Primary Care, and supported by AIDS Prevention Initiative in Nigeria (APIN). FMCA is a 1000-bedded regional specialist hospital that has a goal of providing excellent medical services to Ogun state and other neighbouring states.[21] Ogun state has an HIV prevalence of 1.6% [22] and has 327 centres for the management of HIV.[21]
Study Design
The study was a 3-year retrospective cross-sectionalin design. The study was designed to assess all patients who attended clinic between January, 2021 to December, 2023 and were between the ages 0-9 years and 10-19 years with viral loadeither ≥ 1000 copies/ml.
Record in the HIV Clinic showed that a total population of four hundred and five (405) clients were enrolled for treatment within the period under study, however, one hundred and thirty-five (135, 33.3%) clients were unsuppressed in the period covered by the study. This subset of populationwas subjected to further study and details of the characteristics presented below.
Study population
The study population were individuals between the ages of 0-19 years, who attended HIV clinic between January 2021 and December, 2023 and information captured in the Electronic Medical Records of the hospital. Ethical approval to use the records of the respondents was obtained from the ethical committee of the hospital.
Inclusion criteria
Exclusion criteria
Sampling Technique
The M&E unit of the HIV clinic is saddled with the responsibility of collecting, pooling, and analyses of clinical and epidemiological data gathered by the HIV clinic in conjunction with AIDS Prevention Initiative in Nigeria (APIN) –the Implementing partner with the Federal Medical Centre, in Abeokuta, Ogun State. The dataset stored by the Electronic Medical Records (EMRs) in the M&E unit were accessed, cleaned and analyzed. The EMR database contains data on HIV VL tests performed over the years including the period of investigation with other vital information such as patient’s clinical data and socio-demographic characteristics. Data processes were done as shown in Figure -1 to capture the study period.
Sample size determination
The data set covered a period of 3- years, with the last drug pickup dates ranging from 1st January 2021 to 31st December 2023.
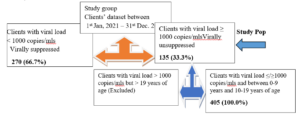
Figure 1- Data cleaning process, excluded data and study population subset analyzed, viral load of respondents.
The minimum sample size was derived from the total number of study population who met the inclusion criteria: children 0–9-year-old with viral load ≥ 1000 copies/ml and adolescent 10-19 years of age with viral load ≥ 1000 copies/ml (unsuppressed viral load within the period of study (January, 2021 to December, 2023) (see table 1 and figure 1) for the breakdown of year-on-yearsampled population.
Data collection
Patients’ data were collected on Federal Ministry of Health approved ART care-cards and subsequently entered into the facility EMR. The data was abstracted from the EMR using an adapted checklist developed from previous local and international studies in related subject.[16, 23, 24] The study population consisted of children from age 0-9 years and adolescent age 10-19 years.[23] A questionnaire was developed to capture and extract patient-level data, containing participant’s specific information including age at time of ART initiation, WHO clinical staging at diagnosis, current ART regimen,most recent viral load results, socio-demographic characteristics of the respondents, and the disclosure status of the child.[23] Patient unique identifiers were excluded from the dataset to ensure anonymity and preserve patient confidentiality. Excel sheets used in the abstraction of study data were encrypted during transmission in line with standardized data safety protocols.
Training of research Assistants and Pre-test
Data collectors and supervisor were trained on the content of the tools, objectives of the study, how to extract the data, how to keep and maintain the confidentiality of the patient data and handle the information they obtained. The checklist was pre-tested at a state-owned health facility before the actual data collection was conducted to ensure the quality of the data. Close supervision by supervisor during data collection was carried out and all data checked for completeness, accuracy and credibility by the principal investigator and other supervisors.
Data Management and Analysis plan
Information extracted from the dataset from the EMR was transferred to an Excel Sheet and same transferred to SPSS version 23 for statistical analysis. Initial analyses were done by generating frequency tables and graphs for categorical variables. Appropriate bivariate analysis was carried out to assess statistical associations depending on the type of the variables and a multivariate logistic regression model was performed to identify predictors of viral non-suppression among children and adolescents age groups.[23] The level of statistical significance was set at p value < 0.05. The adjusted odds ratio and 95% confidence interval were obtained to determine predictors of viral load non-suppression.
Limitation of the study
One of the limitations observed during data collection was the complete absence of parents/care givers information on the Electronic Medical Records for all clients seen at the HIV clinic. It was observed that parents/care givers information were not tagged with the respective clients leading to the inability of this study to provide socio-demographic characteristics of the care givers/parents along with the data for the respondents. A proper triangulation of clients’ data, care givers/parents’ data and clinical environment would help position the institution to deduce the predictors and help improve care of patients.
Socio-demographic characteristics of participants
The socio-demographic characteristics of respondents. The mean age of respondents was 15.76 ± 3.12 and majority 41 (30.4%) were between 14-16 years. It showed that less than half 66 (48.9%) of the respondents were males, while more than half 101 (74.8%) had secondary education. Additionally, (97.8%) of respondents lives in urban area (See Table 1).
Clinical details of respondents
This study had a total population of 405 (100.0%). However, the prevalence of unsuppressed viral load among respondents in this study was 135 (33.3%) between 2021 and 2023. The result of age distribution at diagnosis revealed that the one-third 47 (34.8%) were diagnosed between 0-24 months, followed by 25-60 months 42 (31.1%), and 61-120 months 35 (25.9%). A smaller proportion of patients were diagnosed between 121-160 months 5 (3.7%) and above 160 months 6 (4.4%). Most patients 88 (65.2%) commenced treatment between 0-60 months and majority of patients 109 (80.7%) were diagnosed at Stage I, with fewer cases at Stage II 13 (9.6%), Stage III 6 (4.4%), and Stage IV 7 (5.2%).
Additionally, viral load suppression rates improved over the three-year period. In 2021, about 19 (14.1%) had unsuppressed viral load (Viral load ≥ 1000 copies/ml), while 116 (85.9%) had suppressed viral loads (<1000 copies/ml). In 2022, the proportion of unsuppressed viral load was 12 (8.9%) with increased viral load suppression of 123 (91.1%). In 2023, there was a slight increase in the number of clients with unsuppressed viral load to 16 (11.9%) and 119 (88.1%) were suppressed (See figure 1 and Table 1). Also, the distribution of CD4 counts showed that a significant majority 97 (71.9%) had counts ≥501 cells/µL, while 21 (15.6%) had counts between 351-500 cells/µL, 8 (5.9%) had counts between 201-350 cells/µL, and 9 (6.7%) had counts ≤200 cells/µL.
The therapeutic regimen of the patients
Majority of the client’s 101 (74.81%) were managed with TDF/3TC/EFV, 18 (13.33%) were treated with AZT/3TC/LPV/R and 11 (8.15%) had AZT/3TC/EFV combination, while only 5 (3.70%) were managed with ABC/3TC/LPV/R (See figure 2). Figure 3 – showed that 78% of the participants are compliant with ART, indicating a high level of adherence, while 22% are not compliant. The study revealed that the most common reason for non-compliance is “Skipping medication (41),” followed by “Interruption of treatment (24)” and “Non-compliance with prescription (17).” Other reasons include “Negligence (16),” “Bad adherence (15),” and “Oversight (12).” A smaller number of respondents did not mention their reasons for non-compliance (10) (See Figure 4). Figure 4 illustrates the disclosure status of the participants. According to the chart, 79% of the participants have fully disclosed their status, while 21% have not disclosed their status.
Relationship between viral load Suppression and socio-demographic characteristics/clinical details of respondents
The results from table 3 show that compliance to ART was statistically significantly associated with viral load unsuppressed (Chi-square; P-value-0.025) and disclosure status of respondent (Chi-square; P-value – 0.018) for year 2021. Other factors such as age, gender, education level, location, age at start of treatment, age of diagnosis, therapeutic regimen, CD4 count at diagnosis andWHO staging,were not statistically significant of viral load non-suppression. Similarly, in 2022, compliance to ART was again statistically significantly associated with viral load unsuppressed (Chi-square; P-value-0.015) and CD4 count (Chi-square; P-value- 0.05) of respondents for year 2022. Other factors such as age, gender, education level, location, age at start of treatment, age of diagnosis, therapeutic regimen, disclosure status, WHO stagingwere not statistically significant predictors of viral load non-suppression. For the year 2023, age of respondents at diagnoses (Chi-square; P-value–0.008); compliance to ART (Chi-square; P-value-0.000) and CD4 count (Chi-square; P-value- 0.016) of respondents were all found to be statistically significantly associated with viral load unsuppressed. Other factors such as age, gender, education level, location, age at start of treatment, therapeutic regimen, disclosure status, and WHO staging were all not statistically significant for viral load non-suppression.
Multivariate Analysis of Predictors of Unsuppressed Viral Load among respondents.
The Multivariate regression analysis of predictors of Viral Load Un-suppression among the respondents showed that across the 3-years under review – Compliance to ART was a strong predictor of non-suppression. In the year 2021, Compliance to ART had 2.827 Odd Ratio and P-value- 0.050; in year 2022, the Odd Ratio was 3.811 and P-value – 0.038 and lastly in year 2023, the Odd Ratio was 167.6 and P- value – 0.000 responsible for un-suppression among the respondents (See Table 6).
This study was conducted to determine the prevalence and predictors of viral load non-suppression among children and adolescentsliving with HIV (C/ALHIV) over a 3-year period. Viral load non-suppression remains a daunt task across most HIV programs for C/ALHIV in different parts of the world especially LMICs such as Nigeria and is more obvious when compared to adultclients living with HIV.The mean age of respondents in this study was 15.76±3.12years, majority had secondary level of education, about one-third (34.8%) were diagnosed during the first 2 years of life and only 3.7% were diagnosed after 10years of life. The prevalence of viral non-suppression among the study population was 14.1%, 8.9% and 11.9% for the year 2021-2023 respectively with an average of 11.6% per year. Age at diagnosis, HIV disclosure status and CD4 count were factors significantly associated with viral load non-suppression among the study population. While, age of the patient, gender, educational level, age when ART was commenced, ART regimen and WHO clinical staging were not significantly associated with unsuppressed viral load.
The average overall viral suppression rate of 88.4% documented in this study still falls short of the UNAID’s 3rd 95targeted milestones. The prevalence in our study is higher than the reported values by other researchers across the country [23, 24, 25],and likewise almost triple the national reported value of 34%.26 A nationwide analysis of PLHIV in Nigeriaby Tomescu et al [20]showed that although children in the age group 0-14 years constituted the smallest group (3.4%) among the cohorts but they have the highest percentage (17.8%) of virally unsuppressed individuals.Earlierstudies have shown thatdisparities occur in HIV data both inter- and intra-state and regions in Nigeria [26, 27].Our yearly average of viral suppression rate is comparable with a study conducted in Ethiopia[7]. The viral non-suppression rate in this study is lower than that reported values from some other countries across sub-Saharan Africa (SSA) that ranges between 19.0% to 38.4% [16, 24, 28, 29]The dissimilarities noted may be due to population demographics(social class, education level etc.), study design, study year, ARTtype, duration on treatment as well as differences in programme performance within and across the countries.The recorded highest prevalence noted in our study in 2021 may not be unconnected to the effect of COVID-19 pandemic in 2020 with disruption of many services including health care delivery. The high rate of non-suppression of viral load among C/ALHIV implies that more efforts should be put on interventional strategies such as operation triple zero (OTZ), “test and treat” which may go a long way in the disease management, attain the UNAIDs 95-95-95 targets, improved clinical outcomes and long-term goal of ending AIDS by 2030 [26, 27].
The study showed that poor ART compliance is the most consistent and important contributing factor to unsuppressed viral load among C/ALHIV throughout the study period. This has been acknowledged by earlier authors [21, 24, 25, 30] to be an important contributor to treatment failure and development of drug resistance that may necessitate switching to second- or third-line medications. Some of the cited reasons for poor ART compliance could be pharmacological and/or non-pharmacological. In our study, some of the factors contributing to poor compliance includes skipping medication, interruption in treatment, non-compliance with prescription, oversight, bad adherence and negligence. Aside from these reasons, Aderemi-Williams et al [31]in their study found that negative comment on adolescents’ physical look also has negative impact on their adherence. The finding of poor ART compliance in our study is in tandem with findings from studies conducted in other parts of the country [30]and some other LMICs [28, 32]Children and adolescents living with HIV most often struggle with pills burden (³ 2pills per day), drug-drug interactions, ART side effects, co-morbidities which might contribute to non-compliance with ART. This study also noted that socio-cultural orientation may also have influence on drug compliance as it was observed some parents/caregivers also give native concoctions (herbs)to C/ALHIV. Emphasize on preventive strategies such as enhanced adherence counselling (EAC), reminders via phone calls or SMS to caregivers, tracking, home visits caregivers’ education and support both from healthcare workers, governmental and non-governmental organizations may likely go a long way in overcoming these challenges.
The CD4 count at diagnosis was significantly associated with Viral Load Suppression (VS) and this finding is in tandem with reports from earlier researchers. People living with HIV who had high CD4 count ³ 500 cells/µL have higher VSrate as this reflects restoration of the immune system [4, 25, 33]One other major observation in our study is the low level of disclosure as only 21% of C/ALHIV were aware of their HIV status, this may not be totally unexpected as majority of them were transferred from paediatric clinic. The recommendation by the WHO is that full disclosure should been done by age 12 years for adolescents who acquired HIV perinatally[34]. In spite of WHO recommendation, disclosure rate is still low among C/ALHIV in several countries [31,35]Non-disclosure of HIV in this study negatively correlated with VS, and this is in consonance with observation by other researchers [31, 35].Delayed or lack of disclosure often led to poor adherence among the adolescents as they lack understanding of their disease condition [31]. Major reason cited by Bakai et al [35],and Aderemi-Williams et al [29],that cause non-disclosure of the HIV status to the AYAs were parents’ or guardians’ disapproval and disclosure was even restricted within family members. Therefore, adhering to the WHO recommendation will help in this regard as it has been shown that children with cognitive skills and emotional maturity could handle the information about their HIV status well [35].
Although age at diagnosis of C/ALHIV in this study was not consistent across the years with VS. It became significant in the last year of the study when it was noted that children between 0-24months had a significant risk of VLNS compared to other age categories. Similar observation has been noted by researchers from other SSA [10] Contributory factors include quantity of pills being taken, lack of paediatric formulation, frequency of administration and unpalatable taste of some of the drug e.g. LPV/R. To overcome this challenge, development of paediatric formulation (syrup), reduction of frequency of medications and mother-child linked clinic appointment may likely improve adherence to treatment and VS.
Non-significant factors:
In agreement with other earlies studies [28, 32] our findings imply that there is no significant difference in the risk of VLNS among different age groups. This however contrast with findings by Isaac et al [25]in Northern Nigeriawhere they reported more adolescent were virally unsuppressed compared to children 0-9years. This may not be unconnected to paediatric-friendly services and optimization of ART in operation at our facility.
Our study did not find any association between gender and VLNS, which is similar to findings from Kenya [28] However, there has been varyingreport from within and outside the country. Some authors [10, 20, 25, 33], reported that males are more likely to have virologic failurecompared to females. Reasons adduced for their observationinclude scarce male-friendly healthcare services and poor health seeking behaviour, sex-specific difference in treatment efficacy, toxicity profile and pharmacokinetics [10, 33]
Education status does not influence VS rate our finding which is in contrast to study from rural Uganda where uneducated C/ALHIV with no formal or primary education have higher odds of viral non-suppression [16]. The observe difference may not be unconnected to the fact that almost all the children in the study were in formal education and reside in an urban area. It has been postulated that educated children and adolescents with HIV are more likely to be aware of the significance of ART have compared to the uneducated [26].
Most of the C/ALHIV in this study were started on non-nucleoside reverse transcriptase inhibitor (NNRTI) or nucleoside reverse transcriptase (NRTI) based regimen. Likewise, all the patients in the study were on first-line ART regimen. Nevertheless, these did not affect the viral suppression rate. Our findings on ART regimen did not defers from report from Kenya [28].Though, some authors [25, 28, 30] report higher virologic failure in patients on NNRTI or NRTI.
The current study did not find any association between age at ART initiation and VLNS. There have been contradictory reports on the association between age at initiation and VS. Magedaet al [36] study showed that initiating ART at older age is associated with VLNS and opined that when ART is started early, response to treatment is better as viral replication is still low. Whereas, Muri et al [37] observed that initiating ART at younger age is significantly associated with virologic failure and they suggested that this may possibly be due to subtherapeutic drug levels in younger children, dose-prescribing errors and pharmacokinetics and faster drug metabolism. A well structure paediatric HIV clinic in our facility might have contributed to some of the challenges noted by earlier authors [37]. This study found that there was also no association between WHO staging and VLNS. This may be because majority (> 90%) of our patients are in stage 1 with higher CD4 count at the time of commencement of ART. Earlier study [32] had found that advanced disease stage (WHO stage 3 & 4) is an important risk factor for VLNS.
Table 1: Socio-demographic characteristics and clinical details of respondents
| Variables | Frequency(n= 135) | Percentage (%) |
| Sample population
Suppressed viral load (≤ 1000 copies/ml) Unsuppressed viral load (≥ 1000 copies/ml) |
405
270 135 |
66.7 33.3 |
| Age
Mean = 15.76 ± 3.12 years 5-9 years 10-13 years 14-16 years 17-19 years |
30 39 41 25 |
22.2 28.9 30.4 18.5 |
| Gender
Male Female |
66 69 |
48.9 51.1 |
| Child education level
Primary Secondary Unspecific |
33 101 1 |
24.4 74.8 0.7 |
| Location of abode
Urban Rural |
132 3 |
97.8 2.2 |
| Age at Diagnosis(months)
0-24 25-60 61-120 121-160 >160 |
47 42 35 5 6 |
34.8 31.1 25.9 3.7 4.4
|
| Age at commencement of treatments (months)
0-60 months 60-180 months above 180 |
88 42 5 |
65.2 31.1 3.7 |
| WHO staging at diagnosis
Stage I Stage II Stage III Stage IV |
109 13 6 7 |
80.7 9.6 4.4 5.2 |
| CD4 count(cells/µL)
≤200 201-350 351-500 ≥501 |
9 8 21 97 |
6.7 5.9 15.6 71.9 |
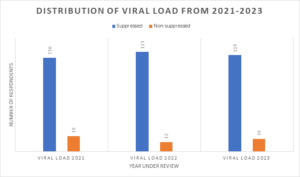
Figure 1: Distribution of viral load from 2021-2023
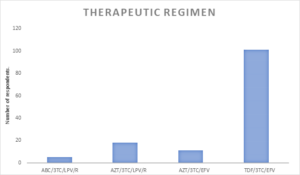
Figure 2: Therapeutic regimen of participants
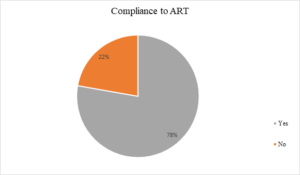
Figure 3: Compliance to ART
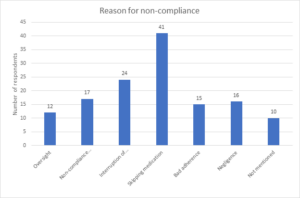
Figure 4: Reason for non-compliance with ART medication
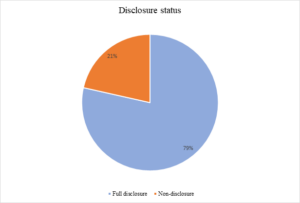
Figure 5: Disclosure status
Table 3: Relationship between viral load and socio-demographic characteristics/clinical details of respondents (2021)
| Viral load 2021 | ||||
| Variables | Suppressed (<1000 copies/mls) | Non-suppressed (≥1000 copies/mls) | X2 -Values | P-value |
| Age of respondents
5-9 years 10-13years 14- 16 years 17- 19 years Total |
22 36 37 21 116 |
8 3 4 4 19 |
5.956 |
.114 |
| Gender
Male Female Total |
53 63 116 |
13 6 19 |
3.376 |
.066 |
| Child education level
Primary Secondary Unspecific Total |
26 89 1 116 |
7 12 0 19 |
1.956 |
.376 |
| Location of abode
Urban Rural Total |
114 2 116 |
18 1 19 |
0.941 |
.332 |
| Age at start of treatment (months)
0-60 months 61-180 months above 181
|
76 35 5 116 |
12 7 0 19 |
1.066 |
.587 |
| Age of diagnosis (months)
0-24 25-60 >60 Total |
37 40 39 116 |
10 2 7 19 |
5.078 |
.079 |
| Therapeutic regimen
ABC/3TC/LPV/R AZT/3TC/LPV/R AZT/3TC/EFV TDF/3TC/EFV Total |
3 15 8 90 116 |
2 3 3 11 19 |
5.310 |
.150 |
| CompliancetoART
Yes No Total |
94 22 116 |
11 8 19 |
5.058 |
.025** |
| Disclosure Status
Full disclosure Non-disclosure Total |
95 21 116 |
11 8 19 |
5.576 |
.018** |
| WHOstaging
Stage I Stage II Stage III Stage IV |
96 9 6 5 116 |
13 4 0 2 19 |
5.611 |
.132
|
| CD4 count (cells/µL)
≤200 201-350 351-500 ≥501 Total |
7 8 19 82 116 |
2 0 2 15 19 |
2.319 |
.509 |
Statistically significant
Table 4: Relationship between viral load and socio-demographic characteristics/clinical details of respondents (2022)
| Viral load 2022 | ||||
| Variables | Suppressed (<1000 copies/mls) | Non-suppressed (≥1000 copies/mls) | X2 – Values | P-value |
| Age of respondents
5-9 years 10-13years 14- 16 years 17- 19 years Total |
25 37 39 22 123 |
5 2 2 3 12 |
4.035 |
.258 |
| Gender
Male Female Total |
60 63 123 |
6 6 12 |
0.007 |
.936 |
| Child education level
Primary Secondary Unspecific Total |
28 94 1 123 |
5 7 0 12 |
2.174 |
.337 |
| Location of abode
Urban Rural Total |
120 3 123 |
12 0 12 |
0.299 |
0.584 |
| Age at start of treatment (months)
0-60 months 61-180 months above 181
|
81 37 5 123 |
7 5 0 12 |
1.054 |
.590 |
| Age of diagnosis (months)
0-24 25-60 >61 Total |
43 39 41 123 |
4 3 5 12 |
0.389 |
.823 |
| Therapeutic regimen
ABC/3TC/LPV/R AZT/3TC/LPV/R AZT/3TC/EFV TDF/3TC/EFV Total |
4 16 9 94 123 |
1 2 2 7 12 |
2.523 |
.471 |
| CompliancetoART
Yes No Total |
99 24 123 |
6 6 12 |
5.880 |
.015**
|
| Disclosure Status
Full disclosure Non-disclosure |
99 24 123 |
7 5 12 |
3.182 |
.074 |
| WHOstaging
Stage I Stage II Stage III Stage IV |
101 10 6 6 123 |
8 3 0 1 12 |
4.391 |
.222 |
| CD4 count (cells/µL)
≤200 201-350 351-500 ≥501 Total |
6 8 19 90 123
|
3 0 2 7 12 |
7.766 |
.050** |
Statistically significant
Table 5: Table 4: Relationship between viral load and socio-demographic characteristics/clinical details of respondents (2023)
| Viral load 2023 | ||||
| Variables | Suppressed (<1000 copies/mls) | Non-suppressed (≥1000 copies/mls) | X2 – Values | P-value |
| Age of respondents
5-9 years 10-13years 14- 16 years 17- 19 years Total |
28 34 35 22 119 |
2 5 6 3 16 |
1.111 |
.774 |
| Gender
Male Female Total |
60 59 119 |
6 10 16 |
0.942 |
.332 |
| Child education level
Primary Secondary Unspecific Total |
31 87 1 119 |
2 14 0 16 |
1.584 |
.453 |
| Location of abode
Urban Rural Total |
116 3 119 |
16 0 16 |
0.413 |
.521 |
| Age at start of treatment (months)
0-60 months 61-180 months above 181
|
78 38 3 119 |
10 4 2 16 |
4.030 |
.133 |
| Age of diagnosis (months)
0-24 25-60 >61 Total |
37 42 30 119 |
10 0 6 16 |
9.706 |
.008** |
| Therapeutic regimen
ABC/3TC/LPV/R AZT/3TC/LPV/R AZT/3TC/EFV TDF/3TC/EFV Total |
5 16 11 87 119 |
0 2 0 14 16 |
2.551 |
.466 |
| CompliancetoART
Yes No |
103 16 119 |
2 14 16 |
44.750
|
0.000** |
| Disclosure Status
Full disclosure Non-disclosure |
92 27 119 |
14 2 16 |
0.868 |
.351 |
| WHOstaging
Stage I Stage II Stage III Stage IV |
99 9 5 6 119 |
10 4 1 1 16 |
5.374 |
.146 |
| CD4 count (cells/µL)
≤200 201-350 351-500 ≥501 Total |
5 7 20 87 119 |
4 1 1 10 16 |
10.386 |
.016** |
Table 6: Multivariate Regression Analysis of Predictors of Unsuppressed Viral Load among respondents.
| Variables | B | S.E. | Sig. | O.R | 95% C.I. for EXP(B) | |
| Lower | Upper | |||||
| 2021 | ||||||
| **Compliance to ART | 1.039 | 0.535 | 0.050 | 2.827 | 0.992 | 8.059 |
| Disclosure status | -1.1 | 0.535 | 0.04 | 0.333 | 0.117 | 0.95 |
| 2022 | ||||||
| **Compliance to ART | 1.338 | .643 | .038 | 3.811 | 1.080 | 13.448 |
| CD4count at diagnosis
(≤ 200) |
.210 | |||||
| CD4count at diagnosis (201- 350) | -20.273 | 13962.026 | .999 | .000 | 0.000 | |
| CD4count at diagnosis (351-500) | -1.598 | 1.064 | .133 | .202 | .025 | 1.629 |
| CD4count at diagnosis (≥501) | -1.779 | .843 | .035 | .169 | .032 | .880 |
| 2023 | ||||||
| **Compliance to ART | 5.122 | 1.219 | 0.000 | 167.617 | 15.36 | 1829.117 |
| CD4count at diagnosis
(≤ 200) |
0.059 | |||||
| CD4count at diagnosis (201- 350) | -1.841 | 2.038 | 0.366 | 0.159 | 0.003 | 8.616 |
| CD4count at diagnosis (351-500) | -5.38 | 2.045 | 0.009 | 0.005 | 0 | 0.254 |
| CD4count at diagnosis (≥501) | -3.672 | 1.547 | 0.018 | 0.025 | 0.001 | 0.528 |
| Age of diagnosis
(0-24 months) |
0.168 | |||||
| Age of diagnosis
(25-60 months) |
-20.091 | 5168.66 | 0.997 | 0 | 0 | . |
| Age of diagnosis
(>60 months) |
-1.75 | 0.926 | 0.059 | 0.174 | 0.028 | 1.067 |
Predictors of Unsuppressed Viral Load among respondents.
Ethics approval and consent to participate
Ethical approval NHREC/08/10/2015 was obtained from the Health Research and Ethics Committee of the Federal Medical Centre,Abeokuta Ogun State; FMCA/470/HREC/01/2024/08. Confidentiality was guaranteed by using numbers rather thannames to identify the information extracted fromeach client. Since participants were no tinterviewed, there was an assurance that thedata collected would be used solely forresearch purposes and there was feedback ofthe findings to the participants.
Consent for publication
All the authors gave consent for the publicationof the work under the Creative Commons Attribution-non-commercial 4.0 license. Weotherwise convey all copyright ownership,including all rights incidental the reto, exclusively to the journal when published.
Availability of data and materials
Data generated in this study are contained inthis manuscript.
Competing interests
The authors have declared that no competing interests exist.
Funding
The research did not receive external funding.
Authors’ contributions
Acknowledgements
We appreciate the healthcare workers who supported us in carrying out this research and for being part of the study.